- Periodic Table of the Elements CHEMISTRY: A Study of Matter. 87 F r (2 3) 55 C s 1 3 2. 9 37 0 Rb 8 5. 1 1 1 N 2 3 3 9 L i 6. 1 H 1 0 1 7 6 2 88 Ra (2 6) 56 B a 1 3 7. 3 38 6 S r 8 7. 20 3 C a 4 0. 1 1 2 M g 2 4. 0 2. 8 9 A c (2 2 7) 5 7 L a 1 3 8. 9. 1 03 L r.
- A site to explain the 2016 OCR A level. Standard Pressure for OCR 100 kPa (kiloPascals) - this isn't precisely 1 atmosphere but previous OCR A levels have always accepted this as an answer.
- This fact-filled, image-rich app is the only periodic table you need. Ideal if you’re a student, teacher or just have an interest in the chemical sciences. Did you know that neodymium is used in microphones? Or europium in Euro bank notes to help stop counterfeiting? These are just two of the absorbing facts in our customisable app, based on our popular and well-respected Royal Society of.
- 3.2.1 Enthalpy changes a) explanation that some chemical reactions are accompanied by enthalpy changes that are exothermic (ΔH, negative) or endothermic (ΔH, positive) Edexcel Chemistry.
- Figure 1: Blank periodic table for students to use in the Introductory Investigation. There is a separate table for each of the four trends, with the headings changed as appropriate. This activity is designed for high school or college general chemistry classes.
A mnemonic is a memory aid used to improve long term memory and make the process of consolidation easier. Many chemistry aspects, rules, names of compounds, sequences of elements, their reactivity, etc., can be easily and efficiently memorized with the help of mnemonics.
Students who demonstrate understanding can use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level of atoms.
*More information about all DCI for HS-PS1 can be found at https://www.nextgenscience.org/dci-arrangement/hs-ps1-matter-and-its-interactions and further resources at https://www.nextgenscience.org.
Contents.History Key dates. Macworld Australia's Commercial Director was Joanne Davies and Editor was Jonathan Stewart. 1985 February–March, premier issue.
Summary: Students who demonstrate understanding can use the periodic table as a model to predict the relative properties of elements based on the patterns of electrons in the outermost energy level of atoms.
Rachel Tarantino (BSU) def. Annie Fidelia (UMB) 6-0, 6-0Doubles competition1. Alisha Silva/Amanda Strickland (BSU) def. Fidelia 1.6.0. Meghan Erickson (UMB) 6-0, retired6. Kristy Garcia/Katie Gauthier (UMB) 8-52.
Assessment Boundary: 
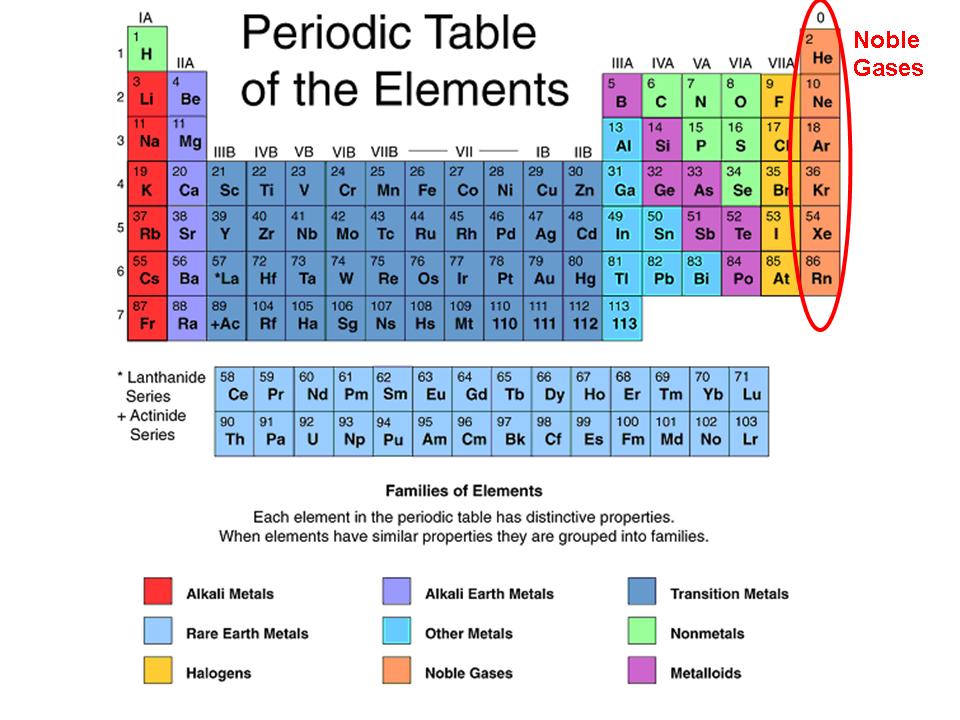
Assessment is limited to main group elements. Assessment does not include quantitative understanding of ionization energy beyond relative trends.
Clarification: Examples of properties that could be predicted from patterns could include reactivity of metals, types of bonds formed, numbers of bonds formed, and reactions with oxygen. Adobe photoshop v20.
Periodic Table Chemistry 3.2.1 Printable
Links:
